November 6


References:

by John Connor
One of the most common question I'm asked is "what should I do for a first cycle?" The questions are; "what steroid should I take?" "How long should I take it?" and "What will the effects be?" There are literally dozens of steroids available and that makes it difficult for a first time user to choose. The following information will attempt to provide enough information for a first time user to make an educated decision about anabolic androgenic steroid use.
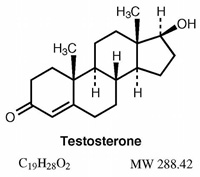
Testosterone is one of the most effective, safe and available steroids today, therefore I believe Testosterone is the best first cycle choice. The following text outlines the benefits and risks of Testosterone administration based on a clinical human trial of 61 healthy men in 2001. The purpose of the trial was to determine the dose dependency of testosterone's effects on fat-free mass and muscle performance. In this trial 61 men, 18-35years old were randomized into 5 groups receiving weekly injections of 25, 50, 125, 300, 600 mg of Testosterone Enanthate for 20 weeks.
They had previous weight-lifting experience and normal T levels. Their nutritional intake was standardized and they did not undertake any strength training during the trial. The only two groups that reported significant muscle building benefits were the 300 and 600 mg groups so any dose lower than 300mg will not be considered in this essay. 12 men participated in the 300 mg group and 13 men in the 600 mg group.

600mg of Testosterone a week for 20 weeks resulted in the following benefits. Increased fat free mass, muscle strength, muscle power, muscle volume, hemoglobin and IGF-1.
The same 600 mg administration resulted in 2 side effects. HDL cholesterol was negatively correlated and 2 men developed acne.
The normal range for total T in men is 241-827 ng/dl according to Labcorp and 260-1000 ng/dl according to Quest Laboratories. The normal range for IGF-1 is 81-225 according to Labcorp. Total T and IGF-1 levels were taken after 16 weeks and resulted in the following;
Total Testosterone
300 mg group-1,345 ng/dl a 691 ng increase from baseline
600 mg group-2,370 ng/dl a 1,737 ng increase from baseline
300 mg group-1,345 ng/dl a 691 ng increase from baseline
600 mg group-2,370 ng/dl a 1,737 ng increase from baseline
IGF-1
300 mg group-388 ng/dl a 74 ng increase from baseline
600 mg group-304 ng/dl a 77 ng increase from baseline
300 mg group-388 ng/dl a 74 ng increase from baseline
600 mg group-304 ng/dl a 77 ng increase from baseline
Body composition was measured after 20 weeks.
Fat Free Mass by underwater weighing
300 mg group-5.2kg (11.4lbs) increase
600 mg group-7.9kg (17.38lbs) increase
300 mg group-5.2kg (11.4lbs) increase
600 mg group-7.9kg (17.38lbs) increase
Fat Mass by underwater weighing
300 mg group-.5kg (1.1lbs) decrease
600 mg group-1.1kg (2.42lbs) decrease
300 mg group-.5kg (1.1lbs) decrease
600 mg group-1.1kg (2.42lbs) decrease
Thigh Muscle Volume
300 mg group-84 cubic centimeter increase
600 mg group-126 cubic centimeter increase
300 mg group-84 cubic centimeter increase
600 mg group-126 cubic centimeter increase
Quadriceps Muscle Volume
300 mg group-43 cubic centimeter increase
600 mg group-68 cubic centimeter increase
300 mg group-43 cubic centimeter increase
600 mg group-68 cubic centimeter increase
Leg Press Strength
300 mg group-72.2kg (158.8lbs) increase
600 mg group-76.5kg (168.3lbs) increase
300 mg group-72.2kg (158.8lbs) increase
600 mg group-76.5kg (168.3lbs) increase
Leg Power
300 mg group-38.6 watt increase
600 mg group-48.1 watt increase
300 mg group-38.6 watt increase
600 mg group-48.1 watt increase
Hemoglobin
300 mg group-6.1 gram per liter increase
600 mg group-14.2 gram per liter increase
300 mg group-6.1 gram per liter increase
600 mg group-14.2 gram per liter increase
Plasma HDL Cholesterol
300 mg group-5.7 mg/dl decrease
600 mg group-8.4 mg/dl decrease
300 mg group-5.7 mg/dl decrease
600 mg group-8.4 mg/dl decrease
Acne
300 mg group-7 of the 12 men developed acne
600 mg group-2 of the 13 men developed acne
300 mg group-7 of the 12 men developed acne
600 mg group-2 of the 13 men developed acne
There were no significant changes in PSA or liver enzymes at any dose up to 600mg. However, long-term effects of androgen administration on the prostate, cardiovascular risk, and behavior are unknown. The study demonstrated that there is a dose dependent relationship with testosterone administration. In other words the more testosterone administered the greater the muscle building effects and potential for side effects.
Given the results of the study and based on years of personal experience I believe the first time user can safely use between 300-600 mg of testosterone enanthate or cypionate per week for 8-12 weeks. Because it is desirable to have even blood androgen levels I advise at least 2 equal injections per week. The following graph demonstrates that testosterone cypionate peaks within 1-2 days after injection and falls off to almost baseline by day 10. Therefore waiting 7 days between injections of cypionate would cause wide fluctuations in blood androgen levels.
Pharmacokinetics of Testosterone cypionate Injection

Source: Schulte-Beerbuhl, 1980 Figure. Pharmacokinetics of 200mg Testosterone cypionate injection. Source: Comparison of Testosterone, dihydrotestosterone, luteinizing hormone, and follicle-stimulating hormone in serum after injection of Testosterone enanthate or Testosterone cypionate. Schulte-Beerbuhl M, Nieschlag E. Fertility and Sterility 33 (1980) 201-3.
If a first time user wanted to use 600 mg of cypionate or enanthate per week he would inject 300 mg on Tuesday and another 300 mg on Saturday each week for 10 weeks. When injecting long heavy esters like cypionate with this frequency I tend to have less acne then 1 injection per week.
There are a number of esters which provide varying release times. Acetate or propionate esters extend the release time of testosterone a couple of days. In contrast, a deconate ester prolongs the release of testosterone about 3 weeks. Testosterone enanthate and cypionate are almost identical esters. The use of an ester allows for a less frequent injection schedule than using a water based testosterone like suspension which has no ester at all and is rapidly in and out of your system after injection. The published release times are not exact and are many times based on a single injection not many multiple injections which can delay the release of the hormone. Other factors affect release times of esters such as scar tissue and the muscle group injected. Only a blood test can confirm when the active hormone has cleared your system.
Esters not only effect release times but also the potency of the Testosterone as esters make up part of the steroid weight. This must be taken into account when calculating dosages. The longer the release time the less free hormone. For example propionate is about 15% more potent mg. for mg. then enanthate so 500mg of propionate would equal about 575 mg. of enanthate. The following chart illustrates the free base equivalents for several compounds.

Although it was not indicated in the trial, during or after the steroid cycle some men are prone to gynecomastia which is the formation of female like breast tissue. This is due to excessive estrogen as the body tries to balance out the sex hormones. A selective estrogen receptor modulator or S.E.R.M. such as Tamoxifen can be used effectively to combat gynecamastia in an emergency as it competes for the estrogen receptor which in turn inhibits estrogen's effects. It is highly recommended that a S.E.R.M. be available during treatment of Testosterone. 10-40mg daily is an effective dose however dosage is dependent on how much testosterone is administered as well as the individual himself.
The decision to use steroids should not be taken lightly and should be the last consideration after implementing a solid nutritional, training and recovery plan. It is advised to get blood work when using these medications.
Ancillaries during the cycle
Aromatase Inhibitor
I briefly wrote about using Tamoxifen above for emergency gynecomastia treatment however I'm convinced that there is a better strategy for controlling estrogen during a steroid cycle. Rather than waiting for the side effects of estrogen to present an aromatase inhibitor like Arimidex or Aromasin should be used on cycle to control Estrogen and keep free testosterone levels high. 0.5mg-1mg Arimidex every other day OR 10-25mg Aromasin daily. Start with the lower dose and then see how that controls water retention, blood pressure and libido and make adjustments as needed. A blood test would be the most ideal way to determine the dosage of the AI. Free T needs to be in the high range and estradiol between 10-30 pg/ml.
Human Chorionic Gonadotropin
Human Chorionic Gonadotropin
Testosterone-Induced gonadotropin suppression tends to cause atrophy of the testes and decreases intratesticular testosterone. In other words, when a male administers testosterone his testes shrink because they are suppressed. A simple way to restore ITT levels and maintain the mass of the testes is to administer HCG during testosterone treatment. During a study it was determined that HCG is dose dependent and that approximately 300iu HCG taken every other day restored ITT levels. This is 1,050iu HCG weekly. I recommend 500iu twice weekly while on testosterone treatment. HCG will not only keep ITT levels and the mass of the testes normal but will also aid in keeping the male fertile.
Sample cycle with ancillaries
Sample cycle with ancillaries
Sunday 10mg Aromasin
Monday 10mg Aromasin/500iu HCG
Tuesday 10mg Aromasin/300mg Enanthate
Wednesday 10mg Aromasin
Thursday 10mg Aromasin
Friday 10mg Aromasin/500iu HCG
Saturday 10mg Aromasin/300mg Enanthate
Monday 10mg Aromasin/500iu HCG
Tuesday 10mg Aromasin/300mg Enanthate
Wednesday 10mg Aromasin
Thursday 10mg Aromasin
Friday 10mg Aromasin/500iu HCG
Saturday 10mg Aromasin/300mg Enanthate
For all you guys who want to add multiple compounds to your first course I advise against it because if you have side effects then you will not know which compound is causing the sides. I have gotten a ton of questions over the years and there is always some reason that I'm given for using multiple compounds on the first run but there really is no need. However my cycle sample above may not be for everyone so I am offering an alternative to the flat cycle design. If you want to run a first cycle with a little more horsepower then you may want to consider a modified pyramiding cycle. I have done over 20 pyramid courses and must say they are my favorite way to run aas. The human body is always fighting for homeostasis so the concept is to increase dose before gains plateau. Based on the 2009 myostatin study we can design a cycle that is effective for 10 weeks using this strategy. The following first cycle is for men that want a little more performance with added risk while only using Testosterone. The first 5 weeks a standard dose is administered to evaluate how your body responds and to determine if sides are manageable. If sides are manageable then increase the dose.
Sample first course #2
Sample first course #2
Week 1-5 600mg Testosterone weekly
Week 6-8 800mg Testosterone weekly
Week 9-10 1 gram Testosterone weekly
Week 6-8 800mg Testosterone weekly
Week 9-10 1 gram Testosterone weekly
10-25 mg Aromasin daily with the goal of keeping Estradiol between 10pg/ml-30pg/ml. Only blood work can confirm if you are in this range.
500iu HCG twice weekly.
Post Cycle therapy
I strongly believe that an AI should be used as long as there is an aromatizing compound being administered. In this case Testosterone and HCG aromatize therefore using an AI until these meds clear is what I'm recommending. Nolvadex has been shown to reduce IGF-1 and GH levels when used alone. This is not a big deal on cycle as testosterone increases IGF-1 in a dose dependent relationship. However off cycle this is may be a problem. PCT is a fragile time and lower IGF-1 and GH levels are not desirable. More advanced users may opt to use Nolvadex and Human Growth Hormone during PCT to counter the HGH lowering effect of Nolvedex. However, I'm recommending AI's that may be used on cycle and during PCT. It's my conclusion that Aromasin or Arimidex are both good choices.
I recommend the following PCT protocol for esters like Cypionate and Enanthate;
While the aas ester is clearing : 2500iu HCG every third day for 2 weeks. (You may use less HCG if your testes are normal in size AND you have been using HCG on cycle, i.e. 1,000iu HCG every third day.)
100/100/100/50 Clomid (50mg taken twice per day weeks 1-3 AFTER the aas ester clears)
20mg/20mg/20mg Aromasin (20mg daily for 3 weeks)
3g Vit C every day split in 3 doses
10g creatine daily
The HCG is administered BEFORE the aas ester clears to increase the mass of the testes and bring back ITT levels. This will allow the testes to sustain output of testosterone sooner.
Clomid is universally accepted as THE testosterone recovery tool. It blocks estrogen from the HPTA and stimulates the production of GNRH then initiates the production of LH, which in turn signals the testis (if not atrophied) to produce testosterone.
Aromasin or a similar aromatase inhibitor is for testosterone recovery and it is used to keep the testosterone/estrogen balance in favor of testosterone. It is also helps to keep any additionally occurring estrogen from HCG low to none.
Cortisol is catabolic. It is the enemy of all anabolism and must be kept in check. While it is blocked when under the influence of AAS, it is free to attach to the Anabolic Receptors (AR) once the steroids leave. Due to this blockage Cortisol tends to accumulate and increase when on. A low level is desirable however since it is important for other vital functions such as control of inflammation. Balance is the key. Vitamin C keeps the exercise induced rise of Cortisol in check.
The use of Creatine has shown to increase ATP metabolism and cellular water storage among many other things. This is beneficial because it provides for heightened nutrient storage and a slight increase in anabolism as well as workout stamina.
Failed Post Cycle TherapySometimes a single post cycle therapy is insufficient to restore healthy testosterone levels and a second post cycle therapy may be needed. In that case I would advise a simple clomid HPTA restart at 50mg daily for 4-6 weeks.
Testosterone dose-response relationships in healthy young men
Shalender Bhasin1, Linda Woodhouse1, Richard Casaburi3, Atam B. Singh1, Dimple Bhasin3, Nancy Berman3, Xianghong Chen4, Kevin E. Yarasheski4, Lynne Magliano2, Connie Dzekov1, Jeanne Dzekov1, Rachelle Bross3, Jeffrey Phillips3, Indrani Sinha-Hikim1, Ruoquing Shen1, and Thomas W. Storer2
Division of Endocrinology, Metabolism, and Molecular Medicine, Charles R. Drew University of Medicine and Science, Los Angeles 90059; 2 Laboratory for Exercise Sciences, El Camino College, and 3 Harbor-University of California Los Angeles Medical Center, Torrance, California 90502; and 4 Biomedical Mass Spectrometric Research Resource, Department of Internal Medicine, Washington University, School of Medicine, St. Louis, Missouri 63110
ABSTRACT
Testosterone increases muscle mass and strength and regulates other physiological processes, but we do not know whether testosterone effects are dose dependent and whether dose requirements for maintaining various androgen-dependent processes are similar. To determine the effects of graded doses of testosterone on body composition, muscle size, strength, power, sexual and cognitive functions, prostate-specific antigen (PSA), plasma lipids, hemoglobin, and insulin-like growth factor I (IGF-I) levels, 61 eugonadal men, 18-35 yr, were randomized to one of five groups to receive monthly injections of a long-acting gonadotropin-releasing hormone (GnRH) agonist, to suppress endogenous testosterone secretion, and weekly injections of 25, 50, 125, 300, or 600 mg of testosterone enanthate for 20 wk. Energy and protein intakes were standardized. The administration of the GnRH agonist plus graded doses of testosterone resulted in mean nadir testosterone concentrations of 253, 306, 542, 1,345, and 2,370 ng/dl at the 25-, 50-, 125-, 300-, and 600-mg doses, respectively. Fat-free mass increased dose dependently in men receiving 125, 300, or 600 mg of testosterone weekly (change +3.4, 5.2, and 7.9 kg, respectively). The changes in fat-free mass were highly dependent on testosterone dose (P = 0.0001) and correlated with log testosterone concentrations (r = 0.73, P = 0.0001). Changes in leg press strength, leg power, thigh and quadriceps muscle volumes, hemoglobin, and IGF-I were positively correlated with testosterone concentrations, whereas changes in fat mass and plasma high-density lipoprotein (HDL) cholesterol were negatively correlated. Sexual function, visual-spatial cognition and mood, and PSA levels did not change significantly at any dose. We conclude that changes in circulating testosterone concentrations, induced by GnRH agonist and testosterone administration, are associated with testosterone dose- and concentration-dependent changes in fat-free mass, muscle size, strength and power, fat mass, hemoglobin, HDL cholesterol, and IGF-I levels, in conformity with a single linear dose-response relationship. However, different androgen-dependent processes have different testosterone dose-response relationships.
Testosterone-induced increase in muscle size in healthy young men is associated with muscle fiber hypertrophy
Indrani Sinha-Hikim1, Jorge Artaza1, Linda Woodhouse1, Nestor Gonzalez-Cadavid1, Atam B. Singh1, Martin I. Lee1, Thomas W. Storer1, Richard Casaburi2, Ruoquing Shen1, and Shalender Bhasin1
1 Division of Endocrinology, Metabolism, and Molecular Medicine, Charles R. Drew University of Medicine and Science, Los Angeles, 90059; and 2 Division of Respiratory and Critical Care Physiology and Medicine, Harbor-University of California at Los Angeles Medical Center, Torrance, California 90509
ABSTRACT
Administration of replacement doses of testosterone to healthy hypogonadal men and supraphysiological doses to eugonadal men increases muscle size. To determine whether testosterone-induced increase in muscle size is due to muscle fiber hypertrophy, 61 healthy men, 18-35 yr of age, received monthly injections of a long-acting gonadotropin-releasing hormone (GnRH) agonist to suppress endogenous testosterone secretion and weekly injections of 25, 50, 125, 300, or 600 mg testosterone enanthate (TE) for 20 wk. Thigh muscle volume was measured by magnetic resonance imaging (MRI) scan, and muscle biopsies were obtained from vastus lateralis muscle in 39 men before and after 20 wk of combined treatment with GnRH agonist and testosterone. Administration of GnRH agonist plus TE resulted in mean nadir testosterone concentrations of 234, 289, 695, 1,344, and 2,435 ng/dl at the 25-, 50-, 125-, 300-, and 600-mg doses, respectively. Graded doses of testosterone administration were associated with testosterone dose and concentration-dependent increase in muscle volume measured by MRI (changes in vastus lateralis volume, http://ajpendo.physiology.org/math/1...rmal/minus.gif4, +7, +15, +32, and +48 ml at 25-, 50-, 125-, 300-, and 600-mg doses, respectively). Changes in cross-sectional areas of both type I and II fibers were dependent on testosterone dose and significantly correlated with total (r = 0.35, and 0.44, P < 0.0001 for type I and II fibers, respectively) and free (r = 0.34 and 0.35, P < 0.005) testosterone concentrations during treatment. The men receiving 300 and 600 mg of TE weekly experienced significant increases from baseline in areas of type I (baseline vs. 20 wk, 3,176 ± 186 vs. 4,201 ± 252 µm2, P < 0.05 at 300-mg dose, and 3,347 ± 253 vs. 4,984 ± 374 µm2, P = 0.006 at 600-mg dose) muscle fibers; the men in the 600-mg group also had significant increments in cross-sectional area of type II (4,060 ± 401 vs. 5,526 ± 544 µm2, P = 0.03) fibers. The relative proportions of type I and type II fibers did not change significantly after treatment in any group. The myonuclear number per fiber increased significantly in men receiving the 300- and 600-mg doses of TE and was significantly correlated with testosterone concentration and muscle fiber cross-sectional area. In conclusion, the increases in muscle volume in healthy eugonadal men treated with graded doses of testosterone are associated with concentration-dependent increases in cross-sectional areas of both type I and type II muscle fibers and myonuclear number. We conclude that the testosterone induced increase in muscle volume is due to muscle fiber hypertrophy.
Testosterone nduced muscle hypertrophy is associated with an increase in satellite cell number in healthy young men
Indrani Sinha-Hikim,1 Stephen M. Roth,2 Martin I. Lee,1 and Shalender Bhasin1
1Division of Endocrinology, Metabolism, and Molecular Medicine, Charles R. Drew University of Medicine and Science, Los Angeles, California 90059; and 2Department of Human Genetics, University of Pittsburgh, Pittsburgh, Pennsylvamia 15261
Submitted 22 August 2002 ; accepted in final form 26 March 2003
Submitted 22 August 2002 ; accepted in final form 26 March 2003
ABSTRACT
Testosterone (T) supplementation in men induces muscle fiber hypertrophy. We hypothesized that T-induced increase in muscle fiber size is associated with a dose-dependent increase in satellite cell number. We quantitated satellite cell and myonuclear number by using direct counting and spatial orientation methods in biopsies of vastus lateralis obtained at baseline and after 20 wk of treatment with a gonadotropin-releasing hormone agonist and a 125-, 300-, or 600-mg weekly dose of T enanthate. T administration was associated with a significant increase in myonuclear number in men receiving 300- and 600-mg doses. The posttreatment percent satellite cell number, obtained by direct counting, differed significantly among the three groups (ANCOVA P < 0.000001); the mean posttreatment values (5.0 and 15.0%) in men treated with 300- and 600-mg doses were greater than baseline (2.5 and 2.5%, respectively, P < 0.05 vs. baseline). The absolute satellite cell number measured by spatial orientation at 20 wk (1.5 and 4.0/mm) was significantly greater than baseline (0.3 and 0.6/mm) in men receiving the 300- and 600-mg doses (P < 0.05). The change in percent satellite cell number correlated with changes in total (r = 0.548) and free T concentrations (r = 0.468). Satellite cell and mitochondrial areas were significantly higher and the nuclear-to-cytoplasmic ratio lower after treatment with 300- and 600-mg doses. We conclude that T-induced muscle fiber hypertrophy is associated with an increase in satellite cell number, a proportionate increase in myonuclear number, and changes in satellite cell ultrastructure.
Androgen Receptor in Human Skeletal Muscle and Cultured Muscle Satellite Cells: Up-Regulation by Androgen Treatment
Indrani Sinha-Hikim, Wayne E. Taylor, Nestor F. Gonzalez-Cadavid, Wei Zheng and Shalender Bhasin
Division of Endocrinology, Metabolism, and Molecular Medicine, Charles R. Drew University of Medicine and Science, Los Angeles, California 90059
Address all correspondence and requests for reprints to: Shalender Bhasin, M.D., Charles R. Drew University of Medicine and Science, 1731 East 120th Street, Los Angeles, California 90059. E-mail: sbhasin@ucla.edu.
Abstract
Androgens stimulate myogenesis, but we do not know what cell types within human skeletal muscle express the androgen receptor (AR) protein and are the target of androgen action. Because testosterone promotes the commitment of pluripotent, mesenchymal cells into myogenic lineage, we hypothesized that AR would be expressed in mesenchymal precursor cells in the skeletal muscle. AR expression was evaluated by immunohistochemical staining, confocal immunofluorescence, and immunoelectron microscopy in sections of vastus lateralis from healthy men before and after treatment with a supraphysiological dose of testosterone enanthate. Satellite cell cultures from human skeletal muscle were also tested for AR expression. AR protein was expressed predominantly in satellite cells, identified by their location outside sarcolemma and inside basal lamina, and by CD34 and C-met staining. Many myonuclei in muscle fibers also demonstrated AR immunostaining. Additionally, CD34+ stem cells in the interstitium, fibroblasts, and mast cells expressed AR immunoreactivity. AR expression was also observed in vascular endothelial and smooth muscle cells. Immunoelectron microscopy revealed aggregation of immunogold particles in nucleoli of satellite cells and myonuclei; testosterone treatment increased nucleolar AR density. In enriched cultures of human satellite cells, more than 95% of cells stained for CD34 and C-met, confirming their identity as satellite cells, and expressed AR protein. AR mRNA and protein expression in satellite cell cultures was confirmed by RT-PCR, reverse transcription and real-time PCR, sequencing of RT-PCR product, and Western blot analysis. Incubation of satellite cell cultures with supraphysiological testosterone and dihydrotestosterone concentrations (100 nM testosterone and 30 nM dihydrotestosterone) modestly increased AR protein levels. We conclude that AR is expressed in several cell types in human skeletal muscle, including satellite cells, fibroblasts, CD34+ precursor cells, vascular endothelial, smooth muscle cells, and mast cells. Satellite cells are the predominant site of AR expression. These observations support the hypothesis that androgens increase muscle mass in part by acting on several cell types to regulate the differentiation of mesenchymal precursor cells in the skeletal muscle.
Effects of testosterone supplementation on skeletal muscle fiber hypertrophy and satellite cells in community-dwelling older men.
Sinha-Hikim I, Cornford M, Gaytan H, Lee ML, Bhasin S.
Division of Endocrinology, Metabolism, and Molecular Medicine, Charles R. Drew University, Los Angeles, CA 90059, USA.
Abstract
OBJECTIVE: In this study, we determined the effects of graded doses of testosterone on muscle fiber cross-sectional area (CSA) and satellite cell number and replication in older men.
PARTICIPANTS: Healthy men, 60-75 yr old, received a long-acting GnRH agonist to suppress endogenous testosterone production and 25, 50, 125, 300, or 600 mg testosterone enanthate im weekly for 20 wk.
METHODS: Immunohistochemistry, light and confocal microscopy, and electron microscopy were used to perform fiber typing and quantitate myonuclear and satellite cell number in vastus lateralis biopsies, obtained before and after 20 wk of treatment.
RESULTS: Testosterone administration in older men was associated with dose-dependent increases in CSA of both types I and II fibers. Satellite cell number increased dose dependently at the three highest doses (3% at baseline vs. 6.2, 9.2, and 13.0% at 125, 300, and 600 mg doses, P < 0.05). Testosterone administration was associated with an increase in the number of proliferating cell nuclear antigen+ satellite cells (1.8% at baseline vs. 3.9, 7.5, and 13% at 125, 300, and 600 mg doses, P < 0.005). The expression of activated Notch, examined only in the 300-mg group (baseline, 2.3 vs. 9.0% after treatment, P < 0.005), increased in satellite cells after testosterone treatment. The expression of myogenin (baseline, 6.2 vs. 20.7% after treatment, P < 0.005), examined only in the 300-mg group, increased significantly in muscle fiber nuclei after testosterone treatment, but Numb expression did not change.
RESULTS: Testosterone administration in older men was associated with dose-dependent increases in CSA of both types I and II fibers. Satellite cell number increased dose dependently at the three highest doses (3% at baseline vs. 6.2, 9.2, and 13.0% at 125, 300, and 600 mg doses, P < 0.05). Testosterone administration was associated with an increase in the number of proliferating cell nuclear antigen+ satellite cells (1.8% at baseline vs. 3.9, 7.5, and 13% at 125, 300, and 600 mg doses, P < 0.005). The expression of activated Notch, examined only in the 300-mg group (baseline, 2.3 vs. 9.0% after treatment, P < 0.005), increased in satellite cells after testosterone treatment. The expression of myogenin (baseline, 6.2 vs. 20.7% after treatment, P < 0.005), examined only in the 300-mg group, increased significantly in muscle fiber nuclei after testosterone treatment, but Numb expression did not change.
CONCLUSIONS: Older men respond to graded doses of testosterone with a dose-dependent increase in muscle fiber CSA and satellite cell number. Testosterone-induced skeletal muscle hypertrophy in older men is associated with increased satellite cell replication and activation.
PMID: 16705073 [PubMed - indexed for MEDLINE]
Adverse effects of testosterone therapy in adult men: a systematic review and meta-analysis.
Ferna¡ndez-Balsells MM, Murad MH, Lane M, Lampropulos JF, Albuquerque F, Mullan RJ, Agrwal N, Elamin MB, Gallegos-Orozco JF, Wang AT, Erwin PJ, Bhasin S, Montori VM.
Knowledge and Encounter Research Unit, Mayo Clinic, Rochester, Minnesota 55905, USA.
Abstract
CONTEXT: The risks of testosterone therapy in men remain poorly understood.
OBJECTIVE: The aim of this study was to conduct a systematic review and meta-analyses of testosterone trials to evaluate the adverse effects of testosterone treatment in men.
DATA SOURCES: We searched MEDLINE, EMBASE, and Cochrane CENTRAL from 2003 through August 2008. Review of reference lists and contact with experts further identified candidate studies.
STUDY SELECTION: Eligible studies were comparative, randomized, and nonrandomized and reported the effects of testosterone on outcomes of interest (death, cardiovascular events and risk factors, prostate outcomes, and erythrocytosis). Reviewers, working independently and in duplicate, determined study eligibility.
DATA EXTRACTION: Reviewers working independently and in duplicate determined the methodological quality of studies and collected descriptive, quality, and outcome data.
DATA SYNTHESIS: The methodological quality of the 51 included studies varied from low to medium, and follow-up duration ranged from 3 months to 3 yr. Testosterone treatment was associated with a significant increase in hemoglobin [weighted mean difference (WMD), 0.80 g/dl; 95% confidence interval (CI), 0.45 to 1.14] and hematocrit (WMD, 3.18%; 95% CI, 1.35 to 5.01), and a decrease in high-density lipoprotein cholesterol (WMD, -0.49 mg/dl; 95% CI, -0.85 to -0.13). There was no significant effect on mortality, prostate, or cardiovascular outcomes.
CONCLUSIONS: The adverse effects of testosterone therapy include an increase in hemoglobin and hematocrit and a small decrease in high-density lipoprotein cholesterol. These findings are of unknown clinical significance. Current evidence about the safety of testosterone treatment in men in terms of patient-important outcomes is of low quality and is hampered by the brief study follow-up.
PMID: 20525906 [PubMed - indexed for MEDLINE]
Pharmacokinetic properties of testosterone propionate in normal men.
Fujioka M, Shinohara Y, Baba S, Irie M, Inoue K.
Abstract
The pharmacokinetic characteristics of testosterone propionate were studied in normal men after a single im dose of 25 mg testosterone propionate-19,19,19-d3. Plasma levels of testosterone propionate-19,19,19-d3, its active metabolite testosterone-19,19,19-d3, and endogenous testosterone were measured by gas chromatography-mass spectrometry. Testosterone propionate-19,19,19-d3 was gradually transferred from the im injection site to the systemic circulation. The plasma levels of testosterone propionate-19,19,19-d3 were maintained at 2-4 ng/ml between 3 and 36 h after administration. Plasma testosterone-19,19,19-d3 levels were maintained above the physiological testosterone level for 48 h, while plasma levels of endogenous testosterone changed little.
PMID: 3782423 [PubMed - indexed for MEDLINE]
The Effects of Injected Testosterone Dose and Age on the Conversion of Testosterone to Estradiol and Dihydrotestosterone in Young and Older Men
Kishore M. Lakshman, Beth Kaplan, Thomas G. Travison, Shehzad Basaria, Philip E. Knapp, Atam B. Singh, Michael P. LaValley, Norman A. Mazer 1 and Shalender Bhasin 1
ivision of Endocrinology, Diabetes, and Nutrition, and Boston Claude D. Pepper Older Americans Independence Center for Function Promoting Therapies (K.M.L., T.G.T., S.Ba., P.E.K., S.Bh.), Boston University School of Medicine, Boston Medical Center, Boston, Massachusetts 02118; Lahey Clinic Medical Center (B.K.), Burlington, Massachusetts 01805; Charles Drew University (A.B.S.), Los Angeles, California 90059; Department of Biostatistics (T.G.T., M.P.L.), Boston University School of Public Health, Boston, Massachusetts 02118; and Hoffman LaRoche (N.A.M.), CH-4070 Basel, Switzerland
Address all correspondence and requests for reprints to: Kishore M. Lakshman, M.D., M.P.H., Division of Endocrinology, Diabetes and Nutrition, Boston University School of Medicine, Boston Medical Center, 670 Albany Street, Second Floor, Boston, Massachusetts 02118. E-mail: Kishore.Lakshman@bmc.org.
Abstract
Background: During testosterone (T) therapy, T is partly converted to 17β-estradiol (E2) and 5α-dihydrotestosterone (DHT). Effects of age, testosterone dose, and body composition on total and free E2 and DHT levels are unknown.
Objective: We evaluated age and dose-related differences in E2 and DHT levels in response to graded doses of testosterone enanthate in young and older men.
Methods: Fifty-one young (aged 19–35 yr) and 52 older (aged 59–75 yr) men completed treatment with monthly injections of a GnRH agonist plus randomly assigned weekly doses of testosterone enanthate (25, 50, 125, 300, or 600 mg) for 5 months.
Results: During testosterone administration, total and free E2 levels increased dose-dependently (dose effect, P < 0.001) in both young and older men. Total and free E2 levels and E2:T ratios during T administration were higher in older than young men, but age-related differences in free E2 and free E2:T ratios were not significant after adjusting for testosterone levels, percentage fat mass, and SHBG. DHT levels and DHT:T ratios were dose-related but did not differ between young and older men. Mechanistic modeling of free hormone data revealed that the conversions of T to E2 and DHT were both consistent with saturable Michaelis-Menten kinetics. The in vivo Km values were estimated to be 1.83 nm for aromatase and 3.35 nm for 5α-reductase, independent of age. The Vmaxparameter for E2 was 40% higher in older men than younger men, but Vmax for DHT was not significantly different between age groups.
Results: During testosterone administration, total and free E2 levels increased dose-dependently (dose effect, P < 0.001) in both young and older men. Total and free E2 levels and E2:T ratios during T administration were higher in older than young men, but age-related differences in free E2 and free E2:T ratios were not significant after adjusting for testosterone levels, percentage fat mass, and SHBG. DHT levels and DHT:T ratios were dose-related but did not differ between young and older men. Mechanistic modeling of free hormone data revealed that the conversions of T to E2 and DHT were both consistent with saturable Michaelis-Menten kinetics. The in vivo Km values were estimated to be 1.83 nm for aromatase and 3.35 nm for 5α-reductase, independent of age. The Vmaxparameter for E2 was 40% higher in older men than younger men, but Vmax for DHT was not significantly different between age groups.
Conclusions: During im testosterone administration, E2 and DHT levels exhibit saturable increases with dose. The rate of whole body aromatization is higher in older men, partly related to their higher percentage fat mass, SHBG, and testosterone levels.
References:
1.Testosterone dose-response relationships in healthy young men;
2.Pharmacokinetics and Dose Finding of a Potent Aromatase Inhibitor, Aromasin (Exemestane), in Young Males
3.Low-Dose Human Chorionic Gonadotropin Maintains Intratesticular Testosterone in Normal Men with Testosterone-Induced Gonadotropin Suppression
4.Use of clomiphene citrate to reverse premature andropause secondary to steroid abuse.
5.Changes in the Endocrinological Milieu After Clomiphene Citrate Treatment for Oligozoospermia: The Clinical Significance of the Estradiol/Testosterone Ratio as a Prognostic Value
6.Testicular steroidogenesis after human chorionic gonadotropin desensitization in rats.
7.Effect of tamoxifen on GH and IGF-1 serum level in stage I-II breast cancer patients
8.Treatment of gynecomastia with tamoxifen: A double-blind crossover study
9.Role of testosterone/estradiol ratio in predicting the efficacy of tamoxifen citrate treatment in idiopathic oligoasthenoteratozoospermic men.
2.Pharmacokinetics and Dose Finding of a Potent Aromatase Inhibitor, Aromasin (Exemestane), in Young Males
3.Low-Dose Human Chorionic Gonadotropin Maintains Intratesticular Testosterone in Normal Men with Testosterone-Induced Gonadotropin Suppression
4.Use of clomiphene citrate to reverse premature andropause secondary to steroid abuse.
5.Changes in the Endocrinological Milieu After Clomiphene Citrate Treatment for Oligozoospermia: The Clinical Significance of the Estradiol/Testosterone Ratio as a Prognostic Value
6.Testicular steroidogenesis after human chorionic gonadotropin desensitization in rats.
7.Effect of tamoxifen on GH and IGF-1 serum level in stage I-II breast cancer patients
8.Treatment of gynecomastia with tamoxifen: A double-blind crossover study
9.Role of testosterone/estradiol ratio in predicting the efficacy of tamoxifen citrate treatment in idiopathic oligoasthenoteratozoospermic men.